Subgroups according to BASDAI/ASDAS category (baseline)
Por um escritor misterioso
Descrição

Improved clinical, functional and work outcomes in spondyloarthritides during real-life adalimumab treatment in central–eastern Europe
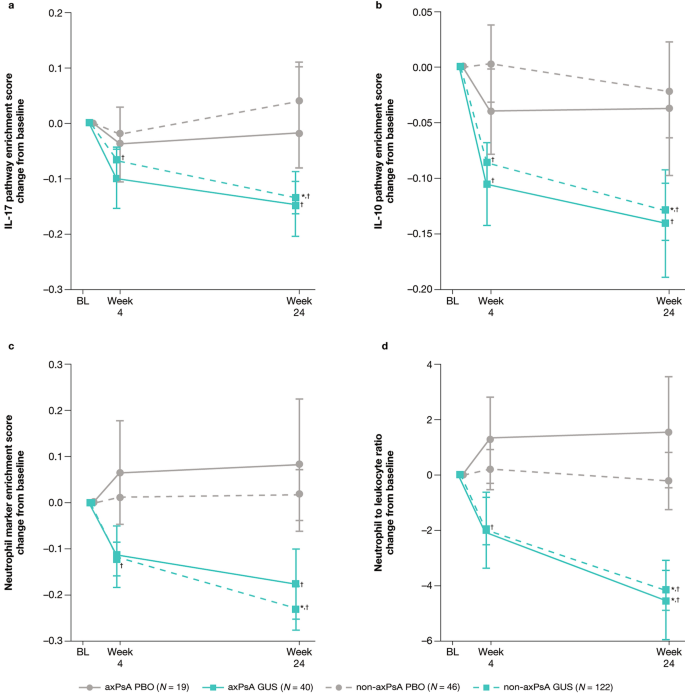
Genetic and Molecular Distinctions Between Axial Psoriatic Arthritis and Radiographic Axial Spondyloarthritis: Post Hoc Analyses from Four Phase 3 Clinical Trials

A and B, Percentages of patients in the golimumab (GLM) and placebo
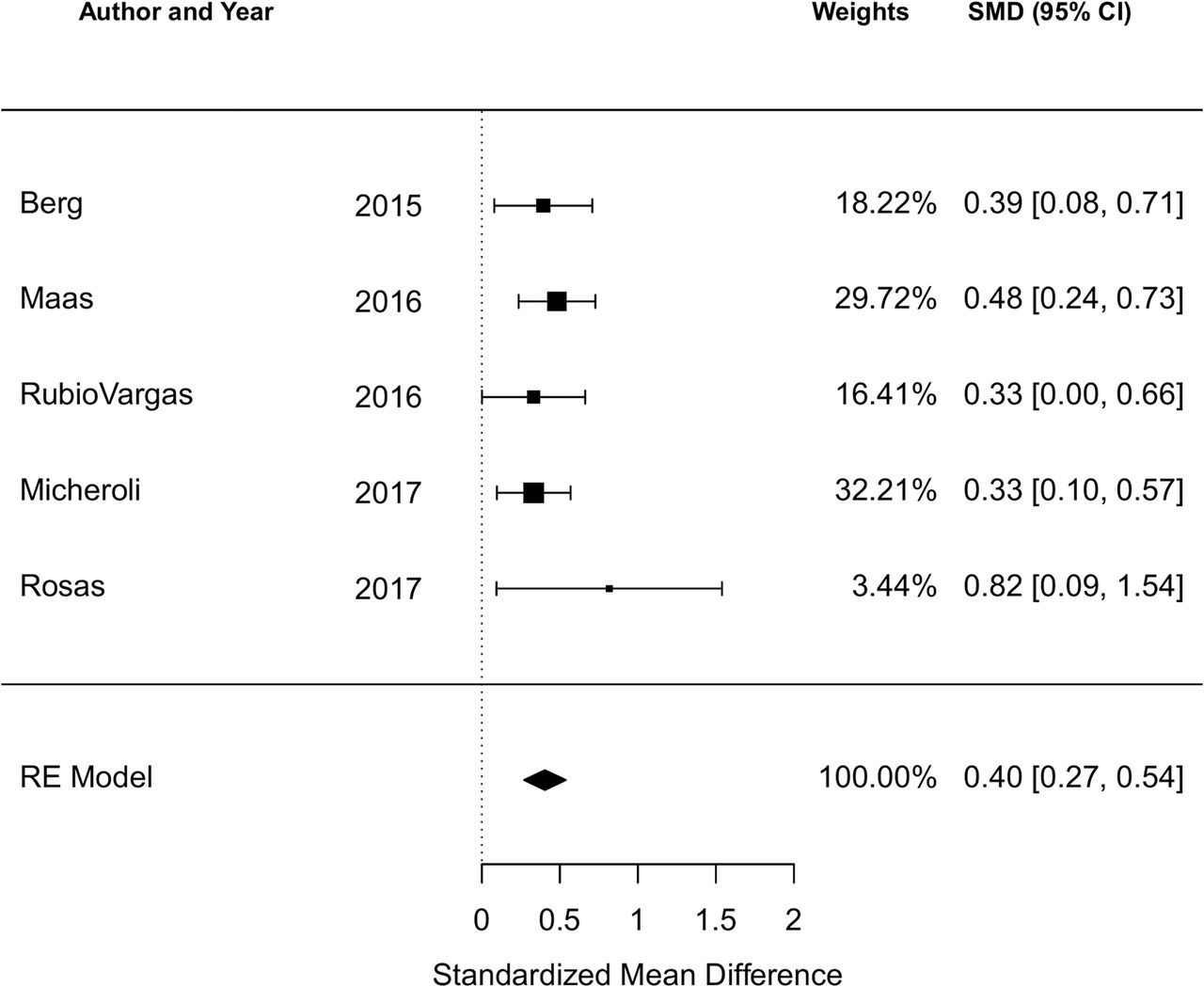
Association of body mass index on disease activity in axial spondyloarthritis: systematic review and meta-analysis
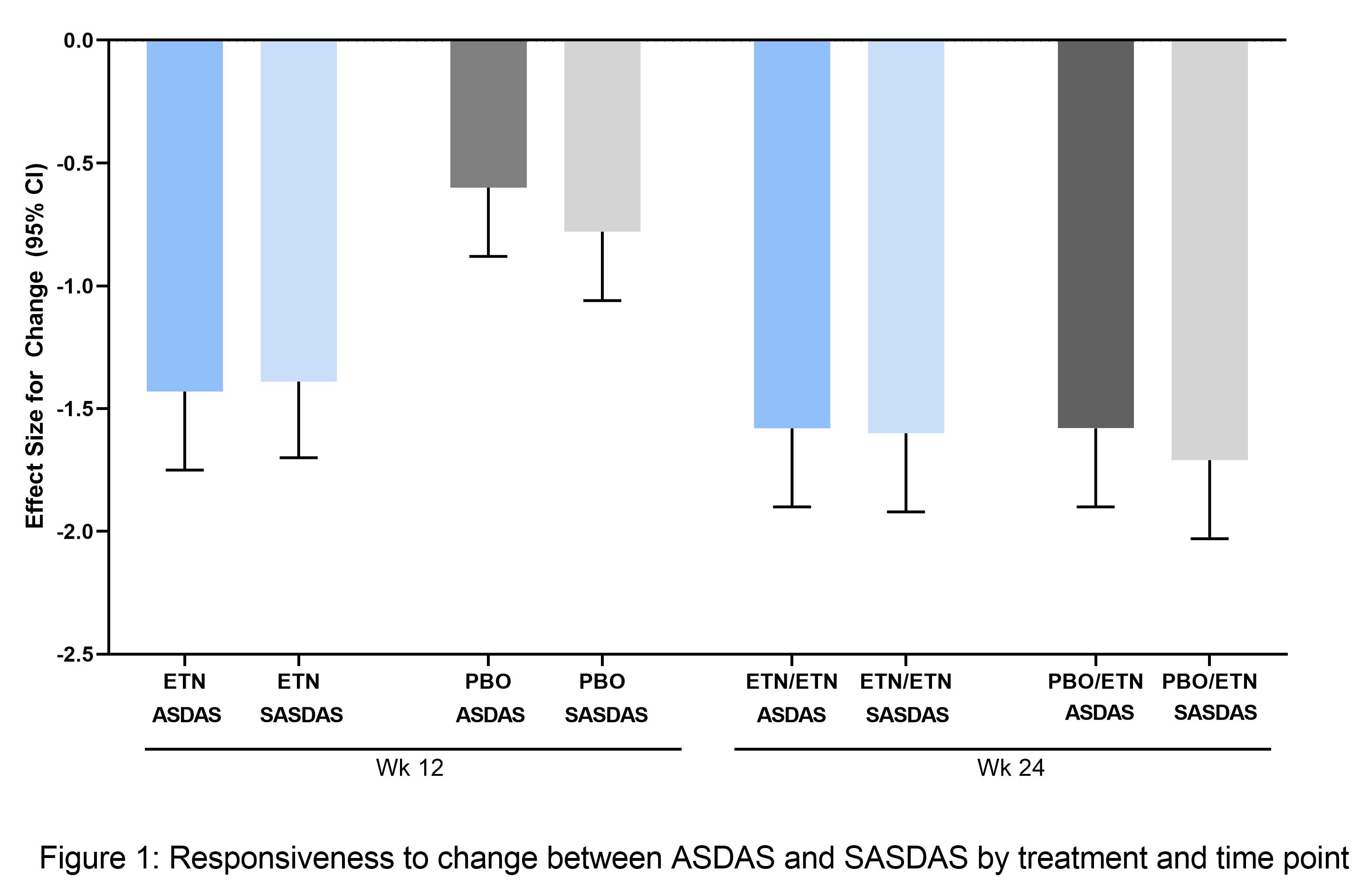
Standardized ASDAS vs standardized SASDAS for (A) baseline pooled data

Efficacy and safety of filgotinib, a selective Janus kinase 1 inhibitor, in patients with active ankylosing spondylitis (TORTUGA): results from a randomised, placebo-controlled, phase 2 trial - The Lancet

NNTs vs. placebo for various thresholds of improvement in BASDAI from

Construct validity and responsiveness of the simplified version of Ankylosing Spondylitis Disease Activity Score (SASDAS) for the evaluation of disease activity in axial spondyloarthritis – topic of research paper in Clinical medicine.

Mean change in modified total Sharp score for C-reactive protein
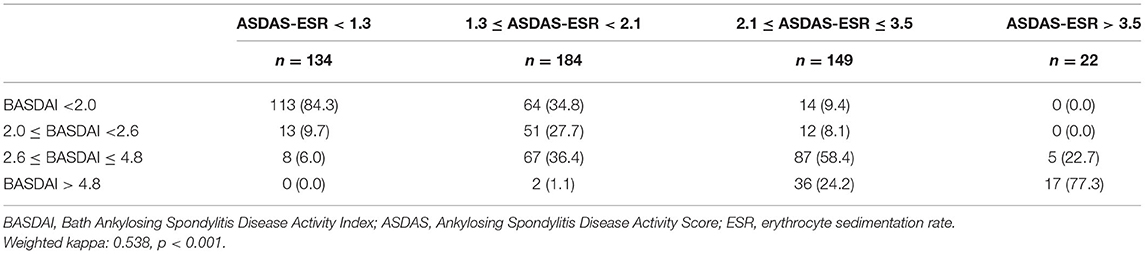
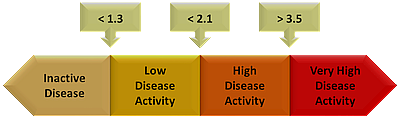
Frontiers The BASDAI Cut-Off for Disease Activity Corresponding to the ASDAS Scores in a Taiwanese Cohort of Ankylosing Spondylitis

Association between baseline fibromyalgianess severity groupings and
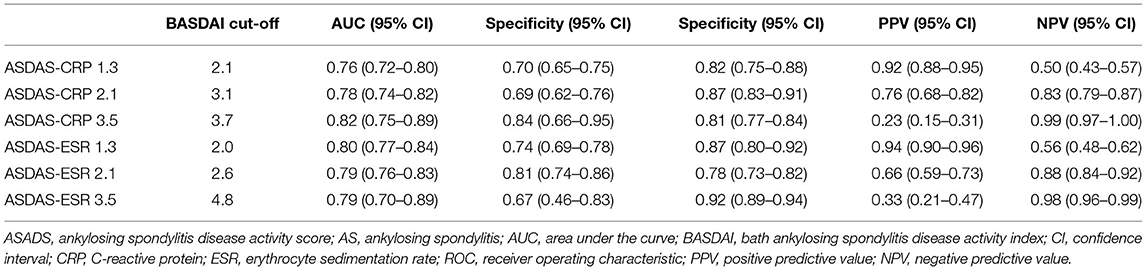
Frontiers The BASDAI Cut-Off for Disease Activity Corresponding to the ASDAS Scores in a Taiwanese Cohort of Ankylosing Spondylitis

Baseline values and 3-month response in subgroups according to CRP

Full article: POSTER PRESENTATIONS
de
por adulto (o preço varia de acordo com o tamanho do grupo)