In Vitro Toxicological Investigation and Risk Assessment of E
Por um escritor misterioso
Descrição
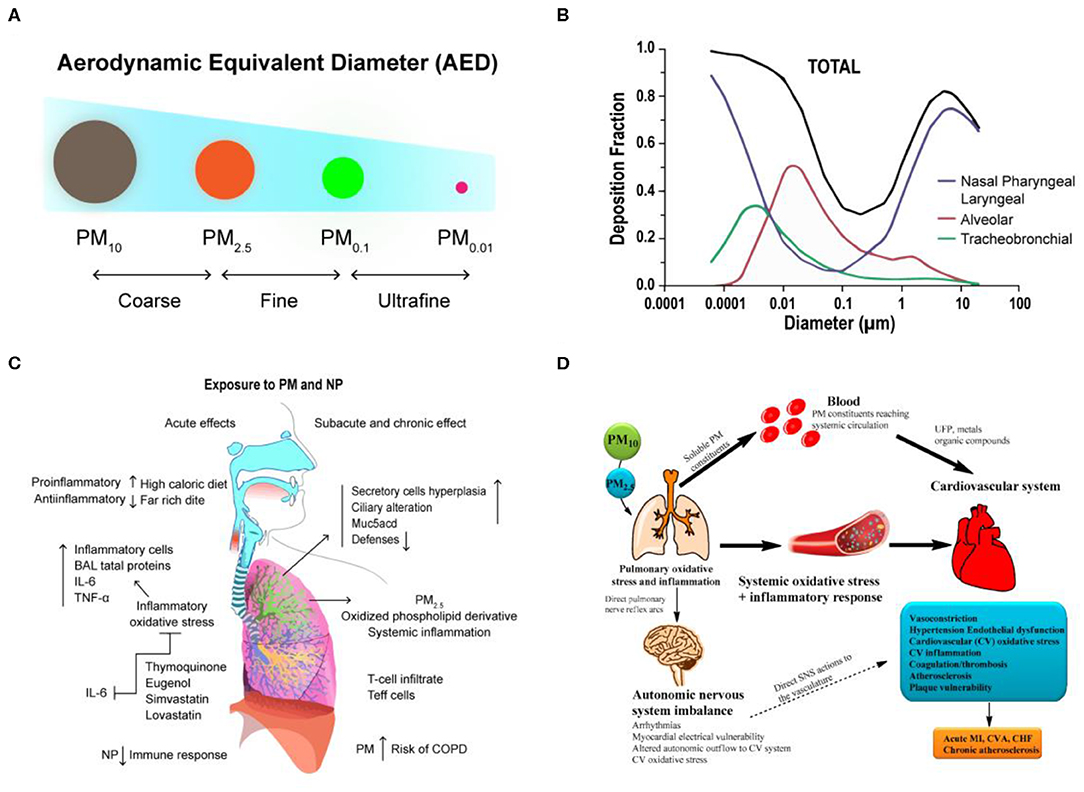
Frontiers Organ-on-a-Chip: Opportunities for Assessing the Toxicity of Particulate Matter

PDF) In Vitro Toxicology, Biodetection and Risk Assessment

Systems Toxicology: From Basic Research to Risk Assessment
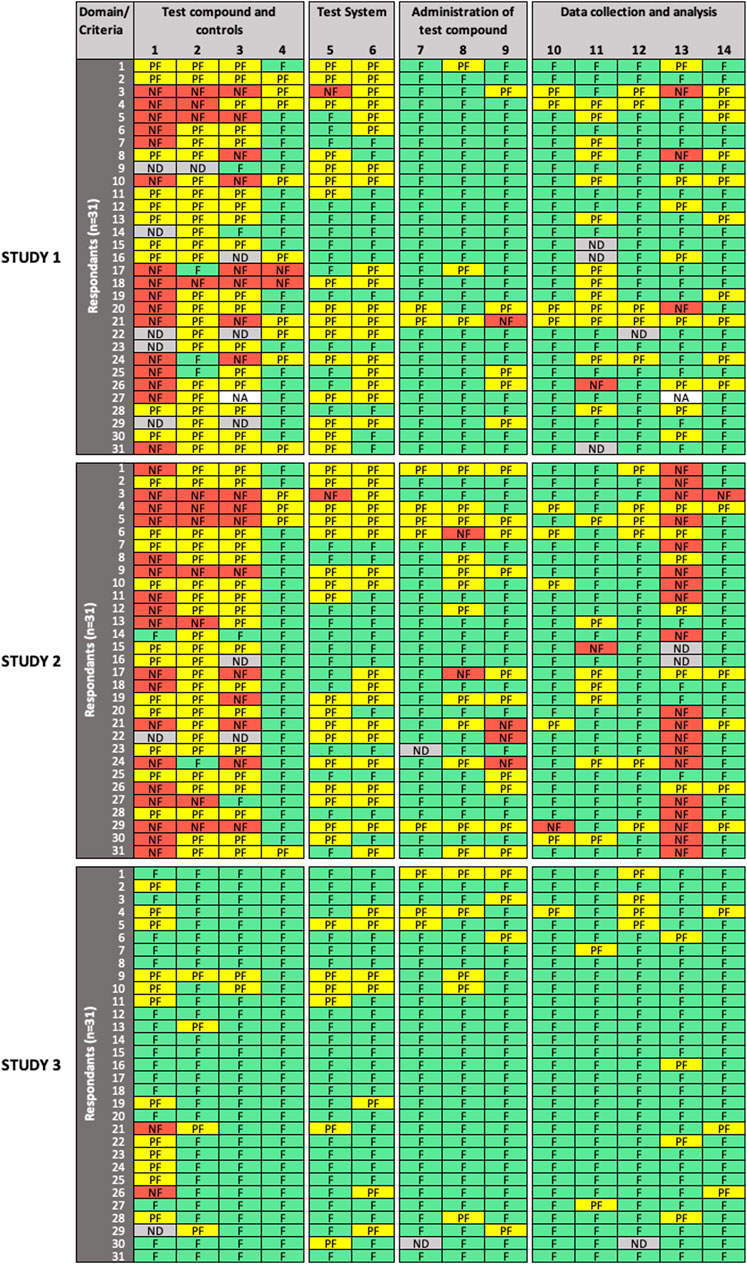
Frontiers Development of the SciRAP Approach for Evaluating the Reliability and Relevance of in vitro Toxicity Data

Toxicological Risk Assessment - an overview

Toxicological Testing: In Vivo and In Vitro Models - ScienceDirect

Guidance on risk assessment of the application of nanoscience and nanotechnologies in the food and feed chain: Part 1, human and animal health - - 2018 - EFSA Journal - Wiley Online Library
Integrated Approaches to Testing and Assessment (IATA) - OECD

Big Data in Chemical Toxicity Research: The Use of High-Throughput Screening Assays To Identify Potential Toxicants

Evaluation of Per- and Polyfluoroalkyl Substances (PFAS) In Vitro Toxicity Testing for Developmental Neurotoxicity
de
por adulto (o preço varia de acordo com o tamanho do grupo)





